What is a Fuel Cell?
A fuel cell is a simple electrochemical device that generates electricity from hydrogen fuel and oxygen.
Fuel cells operate without combustion, so they are virtually pollution free. Since the fuel is converted directly to electricity and heat, a fuel cell’s total system efficiency is far higher than conventional power generation equipment.
Bottom line: Fuel cells are the future of energy.
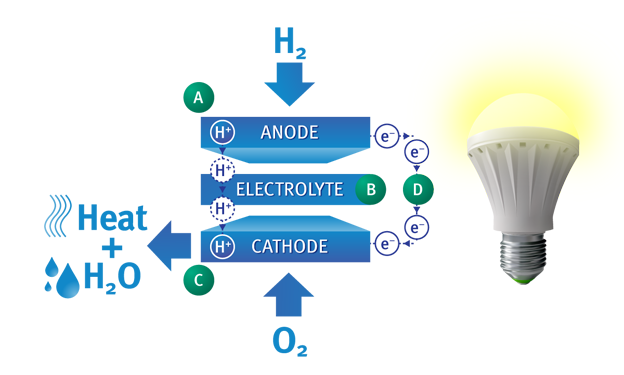
A single fuel cell is composed of an anode (a negative electrode that generates electrons), an electrolyte in the center, and a cathode (a positive electrode that accepts electrons).
Anode
As hydrogen flows into the fuel cell anode, a catalyst layer on the anode helps to separate the hydrogen atoms into protons (hydrogen ions) and electrons.
Electrolyte
The electrolyte in the center allows only the protons to pass through to the cathode side of the fuel cell.
External Circuit
The electrons cannot pass through the electrolyte and are forced to flow through an external circuit. This electron flow is electricity that can power an electric load.
Cathode
As oxygen flows into the fuel cell cathode, another catalyst layer helps the oxygen, protons, and electrons combine to produce pure water and heat.